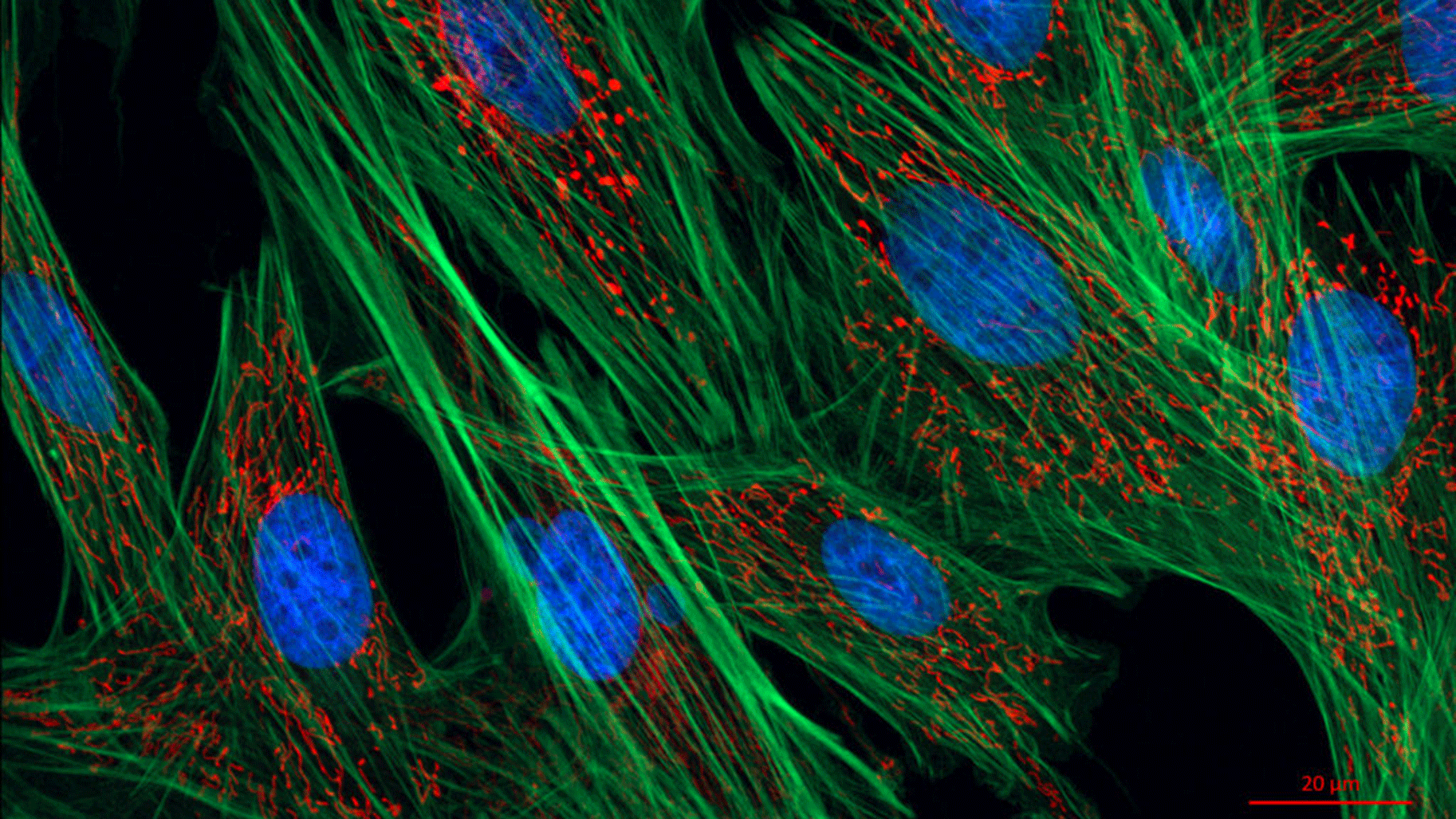
How far may microscopes be able to go one day when it comes to observing the infinitely small? In 1873, physicist Ernst Abbe concluded that the limit was 0.2 micrometres. However, this presumed limitation was exceeded in the 2000s thanks in part to the invention of photoactivatable probes used with fluorescence microscopy, which opened the door to a new world at nano level. Previously, only large components could be observed in cells but now we can also see viruses, proteins and cytoskeletal elements (actin filaments, microtubules, etc.) in detail. This revolutionary development earned its inventors the Nobel Prize in Chemistry in 2014.
It also defined the context in which the Serpico team was born, through a collaborative project between Institut Curie/CNRS (Paris) and the Inria centre in Rennes. Its aim was to design algorithms to process this new crop of images that are often unfortunately blurred, noisy and hard to decipher. In addition to contributing to cancer research, its work also concerns neurobiology, viral and bacterial infections and genetic diseases and opens avenues for the development of more targeted drugs.

Verbatim
We are statisticians, signal and image processors, and machine learning specialists. That is our core field of work. But we work at the interface of a wide range of disciplines and draw on the results of research in biophysics, cell biology, optical technology, machine learning, etc.
head of SAIRPICO team
"In turn, we then produce results that are the fruit of all this work. Our algorithms facilitate the task of fellow researchers such as biologists and microscopists. These tools often address inverse problems, such as restoring images, detecting objects, calculating trajectories and estimating movements. The aim is to facilitate exploration and extract knowledge.”
Multiple collaborations
The scientists’ work is concerned with several imaging techniques, starting of course with fluorescent microscopy, which encompasses multiple directions and involves a range of different partners. “With Institut Curie, we work on exocytosis and endocytosis, which carry molecules out of and into the cell. We want to identify these mechanisms that transport useful molecules such as iron, but are also used by viruses and toxins. Also with Institut Curie, and with the Rennes Genetics and Development Institute (IGDR), we study mitochondria that are at the origin of cancers, genetic diseases and neurodegenerative diseases. With the Institute for Radioprotection and Nuclear Safety (IRSN), we look at problems in detecting and classifying chromosomal aberrations following exposure to excessive radiation. With the Interdisciplinary Institute of Neuroscience (IINS) in Bordeaux, we observe calcium signals in astrocytes [glial cells in the central nervous system]. With the Grenoble Institute of Neuroscience (ING), we characterise the processes of interaction between microtubules and Tau protein, which is found in neurons and is involved in memory. With INRAe, we study the biogenesis of bacteria in order to develop antibiotics for the future. Lastly, with the University of Cambridge, we are looking at transmission factors in order to qualify genetic disorders.”
More generally, the team’s work aims to characterise the dynamics of molecules inside cells over time. “We observe trajectories in very large numbers. These movements can be Brownian - and therefore free - or more directed, or restricted due to molecular congestion. We have also designed something that didn’t previously exist in the literature: statistical tests to categorise and map these movements with a measurement of diffusion at any point in space.”
Electron cryotomography
Another imaging technique is electron cryotomography. “Here, we are no longer in the realm of living matter. Everything takes place in cryogenic conditions. We aim to characterise the spatial organisation and structure of macromolecules of cells. The microscopes used can be several metres tall and the most powerful cost several million euros! They can achieve spatial resolutions of the order of just a few Angstroms, the equivalent of 0.1 nanometres! There are two of these in France and around fifteen in Germany. We work mainly with the Max Planck Institute of Biochemistry (in Martinsried) on the identification of ribosomes. We analyse images for which we need to determine whether we are looking at a membrane ribosome, a cytosolic ribosome, a proteasome or an unidentified macromolecule.” To characterise these elements, the researchers use machine learning techniques based on examples that have already been annotated by biologists: “we have successfully identified macromolecules much smaller than ribosomes, which is a step further than the previous state of the art”.
In terms of the most recent results in cryotomography, the team recently introduced DeepFinder, a deep learning algorithm to identify macromolecules. “There are some 1,000 macromolecules in a cell. In imaging, we can only identify around ten of them. The others are too small. Until now, the literature only allowed us to look for large particles such as 3,200 kDa ribosomes, but with DeepFinder we can descend to 200 kDa, a scale reduction by a factor of 10! This allows us to localise nucleosomes, which are very small particles that form part of the structure of chromatin, an important component of chromosomes.”
The development of these programmes is the result of “often lengthy but necessary dialogue with biologists. They are willing to spend a lot of time with us to design a solution to what is usually a very specific problem. On the other hand, the algorithm we are developing con potentially be used again in many other applications and for other problems in biology.”
BioImageIT
To facilitate the use of these tools, Serpico developed the BioImageIT platform, with the support of the France BioImaging research infrastructure. “This allows biologists to design their own processing pipeline by assembling the components they need via a user-friendly graphic interface. These components can be from any language. The platform works not only with our algorithms but also those developed elsewhere. It is designed to be very inclusive, with the aim of combining the best work in different areas so that everyone can benefit.” BioImageIT has around 6,000 users in France. An engineer travels around the country to install the middleware client and connect the databases of the different establishments, if they exist. Over time, the platform could be extended to other European countries.
Polarised optical microscopy
With the support of the French National Research Agency and within the U1143 Inserm unit, the future Sairpico team will now focus on another imaging technique: polarised microscopy. “The technique has existed for a long time, but in biology it's very new. Mastering it requires a lot of skill.” As for the images produced, “at the moment, nobody really knows how to process them correctly. Very little work has been carried out on this anywhere in the world.”
But what does the technique involve? “Vector images that tell us about the orientation of the molecules of interest at all points in space, or more precisely the orientation of the ‘linker’ which connects the fluorescent molecule to the structure of interest. A colour-coded display gives us an indication of the uncertainty of this orientation, all in 3D+time.” For the layman, the images on the screen look like a collection of overlapping sticks. “As it is, it’s unintelligible. There’s a lot of noise and it’s currently impossible to estimate directionality accurately. The images have to be ordered before any useful information can be extracted from them.”
This work requires collaboration with opticians and biologists, but also chemists. “These are the people who design probes to make the linkers as rigid as possible. If they are too “bendy”, we can’t measure anything of interest.” This is where the collaboration with the chemists and biologists of the Inserm chemical biology unit really comes into its own.
Sairpico joins a chemical biology unit
“The unit focuses on chemical cell biology. It combines basic research in cell biology and chemistry for medical purposes, such as the fight against cancer. The idea is to develop new techniques from basic results in chemistry to target compartments, forms of transport or biomolecules inside tumour cells. This is known as nanotherapy. We need to 'find' ways to get nanodrugs into the cell and deliver them to their destination in a precise manner. Unit 1143 is composed of two teams of biologists and one of chemists. Sairpico will be the fourth team in the Unit and will add to the scope of this project by providing input from computational imaging and artificial intelligence." At the same time, three Inserm researchers are also joining the team.

Know more about BioImageIT with Charles Kervrann (french)