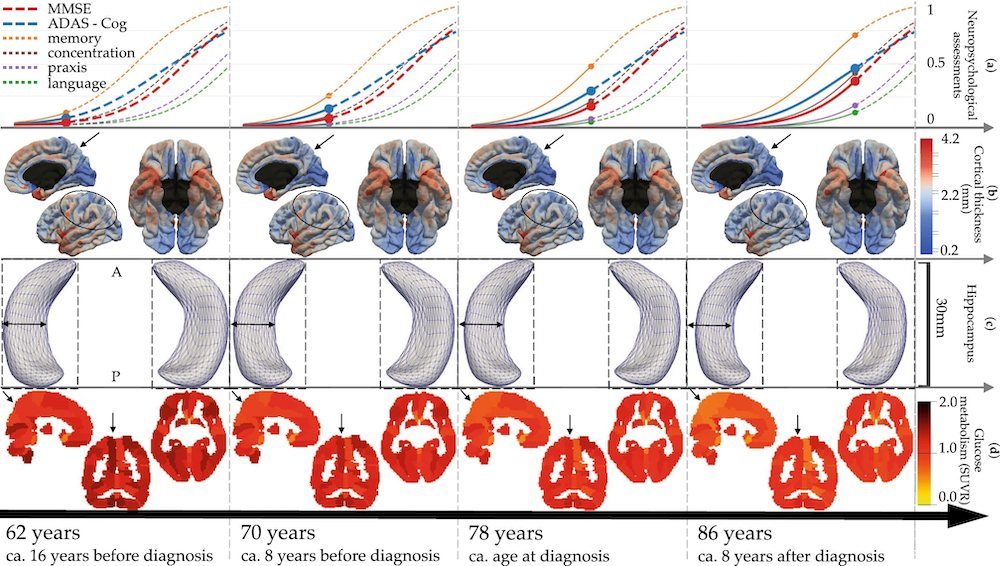
Customizable models of the progression of Alzheimer's disease presented at four different stages with estimated time to diagnosis. The different images (from bottom to top) show the alteration of glucose metabolism in the brain, atrophy of the hippocampus, thinning of the cortex and the onset of cognitive decline. Illustration from Scientific Report, 11, 8020 (2021).
Digital medicine to predict cognitive changes
For the last decade, Stanley Durrleman and his ARAMIS project-team at the Inria Paris centre, a component of the Brain Institute Paris (ICM) working jointly with the CNRS, INSERM and Sorbonne University, have been seeking to predict changes to the brain in patients suffering from neurodegenerative diseases, and they are succeeding! They have created a model, called the AD Course Map, which combines geometry and statistics to predict the course of patients based on their initial medical data.
The model’s effectiveness has been proven through simulations. Imagery results and clinical tests were transferred to the algorithm, which then predicted patients’ cognitive development, producing an outcome closely in line with that observed in reality. AD Course Map also forecasts cognitive decline more accurately than 56 other alternative methods developed as part of the Tadpole Challenge, organised in 2019 by the European initiative, EuroPond, (bringing together computational neuroscientists and clinical neurologists) in collaboration with the Alzheimer’s Disease Neuroimaging Initiative.
Clinical trials prove difficult for Alzheimer’s disease
"It’s great to be able to predict, but to what end?" Stanley Durrleman asks. According to the researcher and his team, there are two leads to follow. The first is to improve patient care by offering a treatment protocol and follow-up adapted to the forecast trajectory of the disease, for example.
The second is to support pharmaceutical research.
There is very little treatment for Alzheimer’s disease on the market and one of the reasons for this is that it is difficult to judge their effectiveness during clinical trials’, the co-director of ARAMIS explains. ‘This disease can remain stable for several years, so the benefit of a molecule may lessen during a trial, because even in the placebo group, there may statistically be very little development.
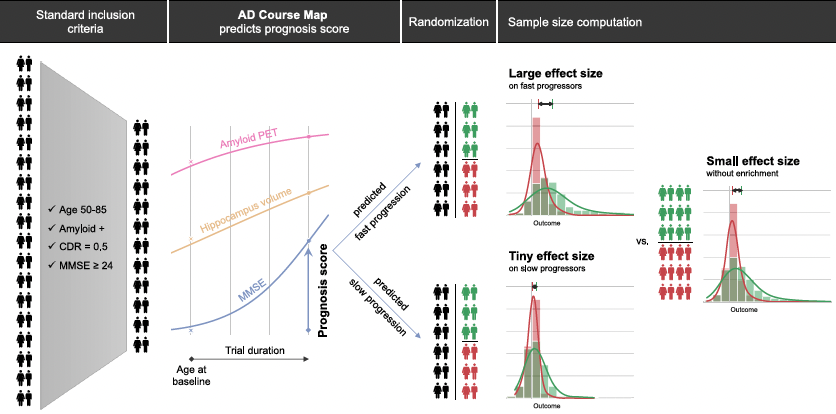
Improving patient selection for more significant results
Nevertheless, if a model can predict when patients will experience cognitive decline, it is then possible to include only such subjects in the trial, and thus measure the effectiveness of treatment in enhanced conditions. The ARAMIS project-team have just demonstrated this fact with the results of their study, published in Nature Communications.
The first step in their research was to train the model using medical data from 80% of patients in a North American cohort presenting biomarkers of Alzheimer’s, but not necessarily diagnosed with the disease. The remaining 20% were used to test the accuracy of AD Course Map predictions. The model was then applied to four other cohorts, in Japan, Europe and Australia, for a total of 4,687 patients who had been monitored for four years.
A model which overcomes geographical and gender bias
The first good news was that even if the training of the model was based on a North-American population, the predictions were as good, if not better, for the other populations; a generalisation which is quite rare in medicine’, Stanley Durrleman points out. The second positive aspect is that the model is not gender-biased, so while the course of the disease generally differs in men and women, the model can forecast their outcomes just as efficiently.
Furthermore, AD Course Map is more efficient than another predictive model, RNN-AD, which is based on deep learning. “These kinds of predictive models require vast amounts of input data to discover their hidden patterns, says the researcher. ‘In the case of Alzheimer’s, data, and more particularly long-term information, is somewhat limited, because patients are rarely followed for more than a few years and the data collected is variable. In addition, deep learning works well for classification, but what we need here is not a binary or even tertiary response, rather an individualised forecast over a relatively long period.”
Which patients for which trials?
The most interesting aspect of AD Course Map is its relevance in the selection of a clinical trial cohort. Drawing on the protocols of six previous trials, the ARAMIS team have proven that the number of patients required to assess the effectiveness of a treatment programme can be reduced by 30% to 50%. “By directly targeting the patients whose outcome corresponds to that which the drug should be treating, we increase the power of the trial,’ Stanley Durrleman explains. ‘We can see a real change in the placebo group patients, whereas the effect of the molecule will be visible, or not, in the treated group.”
These results will further strengthen collaboration between ARAMIS and pharmaceutical laboratories. The latter have already called on INRIA to study the possibility of applying the model to their clinical trials, or to observe the results of previous trials retrospectively. This type of model is gaining ground, in oncology for instance, and the European Drugs Agency has even issued a favourable view for its use in designing clinical trials.
“In the two years preceding our studies, the market launch of the first Alzheimer drug was approved by the American Food & Drug Administration, despite the fact that clinical trials had demonstrated its low effectiveness”, Stanley Durrleman tells us. “This has rightly raised questions, and there is an urgent need to set up clinical trials which will assess the benefit of molecules more efficiently. This will allow us, on one hand, to avoid treating patients for nothing, with sometimes non-negligible side-effects, and on the other hand, limit the costs incurred for the health system.”
Individual prediction for personalised treatment
The researcher also suggests his model could be applied to the treatment of patients. He created a start-up last September to set up a digital clinic, ‘La Clinique du Docteur Mémo’ [‘Dr. Memo’s Clinic’], accessible at docteurmemo.fr (in French). The clinic enables people concerned about their memory faculties to answer a short orientation questionnaire designed by neurologists. Depending on their results, they will be reassured or directed towards a consultation with a doctor trained specifically in the treatment of cognitive disorders.
Using the AD Course Map, this doctor can then suggest appropriate treatment. “A patient who should remain stable for a relatively long period will be given cognitive boosting exercises to do at home, while patients for whom the algorithm predicts a faster decline will benefit from medical treatment and more regular monitoring,” Stanley Durrleman explains.
What is the key purpose of this approach? To put patients in contact with a specialist right from an early stage of the disease. “We know we have to diagnose early, but general practitioners are rarely trained for this reality”, the scientist comments. “How can we ensure early treatment if the diagnosis is held up at the first link of the chain? Our digital clinic will help to by-pass that hurdle.” The task at present is to publicise its existence as widely as possible.
Read the article in Nature Communications:
“Forecasting individual progression trajectories in Alzheimer’s disease”, Maheux, E., Koval, I., Ortholand, J. et al., Nature Communications 14, 761 (2023).
Find out more:
- Neuro-imagerie, neurologie et modèles numériques pour la maladie d'Alzheimer (vidéo), Collège de France, 2/5/2018.
- Des modèles numériques pour prédire et traiter Alzheimer par Stanley Durrleman (vidéo), S3Odéon, 15/2/2018.
- Initial identification of the early signs of Alzheimer’s disease, Inria, 23/2/2022.
- Stanley Durrleman - La modélisation au service de la recherche sur les maladies neurodégénératives, Data Analytics Post, 16/9/2021.