Modelling the real to encompass its full complexity
The human body is complex and variable, which means that some patients respond badly or in a less-than-optimal way to a nevertheless well-established medical protocol. Ideally, by examining each patient in detail prior to any intervention or drug prescription, we could avail of truly patient-specific medical care. While this degree of analysis remains utopian at present, mathematical modelling already enables us to deepen medical understanding by identifying the mechanisms at work in pathologies and virtually testing hypotheses and treatment protocols.
Creating a dialogue between fields of expertise to link micro and macro levels.
Such is the task that Dirk Drasdo and Irene Vignon-Clementel, both senior researchers at the INRIA Saclay Île-de-France Centre, have set themselves. Together, they coordinate SIMBIOTX (SImulations in Medicine, BIOtechnology and ToXicology of multicellular systems). Dirk Drasdo provides the team with expertise in multi-cellular tissue modelling created from image data at tissue level. His quantitative and dynamic models enable monitoring of the spatial and temporal development of systems in vitro and in vivo, from the individual cell to the tissue block. These are combined with macroscopic methods for the simulation of blood flows in vivo, developed by his colleague. The result is hybrid models that contribute to advancing our understanding of the links between events on a microscopic cellular level and the systemic macroscopic consequences.
The two researchers have thus succeeded in demonstrating that we can compare sections of tumour tissue and the data obtained through certain imaging methods. Thanks to the understanding of this relationship, the in silico models will be configured to reflect the architecture, perfusion and function of the organs observed via imaging. Combined with the analysis of various useful biomarkers, this breakthrough could ultimately enable tumour diagnosis by reducing the need for invasive analysis methods, such as biopsies. By furthering the understanding of disease progression, these models will also help to plan appropriate treatment.
Digital twin: In silico checking of scientific hypotheses
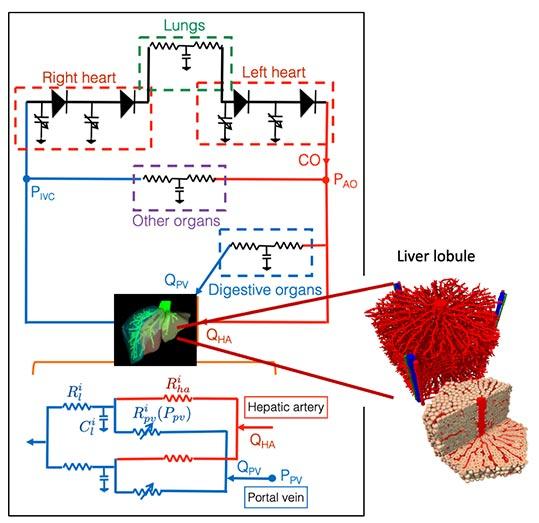
The method developed by SIMBIOTX also enables us to extrapolate in vitro toxicological experiments to humans. The team is working on this question in close collaboration with the toxicology department of the IfADo Leibniz Institute (Germany). Let us consider a case of paracetamol overdose, for example. This causes the death of the hepatocytes surrounding the central vein of each lobule of the liver, followed by their rapid regeneration. By digitally simulating each individual hepatic cell, the team creates a virtual twin of a section of the liver, which helps to determine if and under which conditions hypothetical mechanisms explain observational data (e.g. the dose dependence of the affected area).
If the hypothesis put forward does not match the clinical observation, the models can test alternative mechanisms and contribute to the design of experiments to be carried out on animals in order to obtain confirmation of hypotheses. “Once a model has been configured correctly and validated by data, it can be used to simulate the influence of different disturbances, such as molecular manipulations or the blocking of the expression of certain genes, for which the effects on the organ and the modelled workings are established”, Dirk Drasdo explains. By helping the experimenter to identify which experiments have the greatest influence on their measurements, these models contribute to reducing the number of experiments to carry out, particularly on animals.
Bringing innovation to the operating theatre
In direct response to clinical issues, Irène Vignon-Clementel develops digital tools to try to understand surgical risks, starting with operations on congenital heart diseases. As part of this work, she has already collaborated with Stanford Children’s Hospital (USA), and Necker Children’s Hospital (Paris).
She has shared a common focus with Dirk Drasdo on the liver and liver surgery (resection, transplant, etc.) for several years and their aim now is to generate a digital model of the organ on several levels of organisation, from the individual cell to the working organ, with its lobule architecture and blood flow. “While anatomical models have recently been developed, along with multi-parameter algorithms for decision support, they are not sufficiently accurate to anticipate post-operational effects, because they do not take into account the dynamics of the patient’s blood flow”, she explains.
At SIMBIOTX, and in association with clinicians, she is looking to develop such “haemodynamic” models to predict the response of a specific liver to partial ablation, according to the re-distribution of blood flow and pressure. This is the particular focus of their partnership with the team led by Eric Vibert, Professor of Surgery at Paris-Saclay University, in the Haepatobiliary Centre of the APHP Paul Brousse Hospital.
“By using the power of mathematics to analyse without simplifying the real, this collaboration has enabled us to improve our understanding of haemodynamics. In the operating theatre, this results in improved anticipation of the consequences of our surgical acts and a better selection of operable diseases”, the clinician explains.
Moving towards patient-specific surgery
For the moment, these models are being used retrospectively to understand a posteriori the changes to blood flows triggered by operations. However, Irène Vignon-Clementel hopes that “within the next five to ten years, they will be sufficiently predictive and validated for use by expert clinicians to plan operations”. Furthermore, she received an ERC grant in late 2019 for her MoDeLLiver project to develop a haemodynamic model for the planning of lung and liver surgical operations.
By putting the power of mathematical modelling to work, SIMBIOTX allow the mechanisms which govern the workings of the human body to be identified - from a cellular level to whole organs - and of the blood system irrigating them. Fields of application are also moving into biotechnology and the creation of prostheses, etc. One of the key strengths of these models is that they enable an examination of the complex interaction of tissue and cell biomechanics, biochemistry and flows on several levels. “The downside is that they require the handling of a complex code. If we had more resources, we could do even more and faster, because we are seeing huge interest from clinicians”, Irène Vignon-Clementel adds. Note to project backers...
Find out more about SimbiotX research
- Optimizing cancer drug therapies, INRIA, 08/02/2021.
- Irene Vignon-Clementel & Stéphane Cotin - Le numérique, appui au contrôle de risque en chirurgie [Digital support for surgical risk control], INRIA Foundation, 04/01/2021.
- MOX Colloquium: Irene Vignon-Clementel, “Patient-specific hemodynamics simulations for interventional planning of congenital and acquired cardiac diseases”, Laboratorio MOX, 21/01/2021.
- Digital twins in the health sector: mirage or reality? Institut Mines-Télécom, 28/01/2019.