The EPIMETHEE team from the Decision and Bayesian Computation de l’Institut Pasteur de Paris. From left to right: Jean-Baptiste Masson (project team leader), François Laurent (research engineer), Alexis Bénichou (PhD student), Christian Vestergaard (permanent researcher).
Neuroscience: unlocking the mysteries of neurodegenerative diseases
Fruit flies might be a real a nuisance to greengrocers, but for EPIMETHEE, a new project team launched in 2023 by Inria, the Pasteur Institute and the CNRS, fruit fly larvae are an object of study, and the source of much fascination. By studying their neural circuits, over the next few years the team is hoping to be able to unlock some of the mysteries of the human nervous system, drawing on computational biology. This includes neurodegenerative diseases such as Alzheimer’s and Parkinson’s, as well as inflammatory diseases such as multiple sclerosis.
“We want to explain the impact these conditions have on the structure of our own neural circuits and how they function”, explains Jean-Baptiste Masson, the head of the project team. “We are also seeking to improve the first steps in the testing process for candidate drugs for neurodegenerative diseases.” In turn, this will help to improve how these conditions are treated.
An interdisciplinary team centred around bioinformatics
The EPIMETHEE project team is linked to the Decision and Bayesian Computation laboratory at the Pasteur Institute in Paris, which is also headed up by Jean-Baptiste Masson. The team’s ten members are specialists in theoretical physics, neuroscience, software and Bayesian inference. Further down the line they will be joined by a researcher from Inria, who will contribute their knowledge of theoretical mathematics, simulation and development.
Jean-Baptiste Masson is a theoretical physicist by training, but he is also the co-founder and scientific director of Avatar Medical, a startup that has developed a virtual reality tool to help surgeons prepare for procedures by using medical images to generate 3D representations :
Verbatim
What we want to do is to describe how evolution optimised fruit fly larvae’s decision-making circuits. This will involve bringing multiple disciplines together, including bioinformatics and computational biology, with highly complex models at the intersection of physics and mathematics. Inria’s contribution on this aspect will be vital.
Project team leader
A model more versatile than artificial intelligence
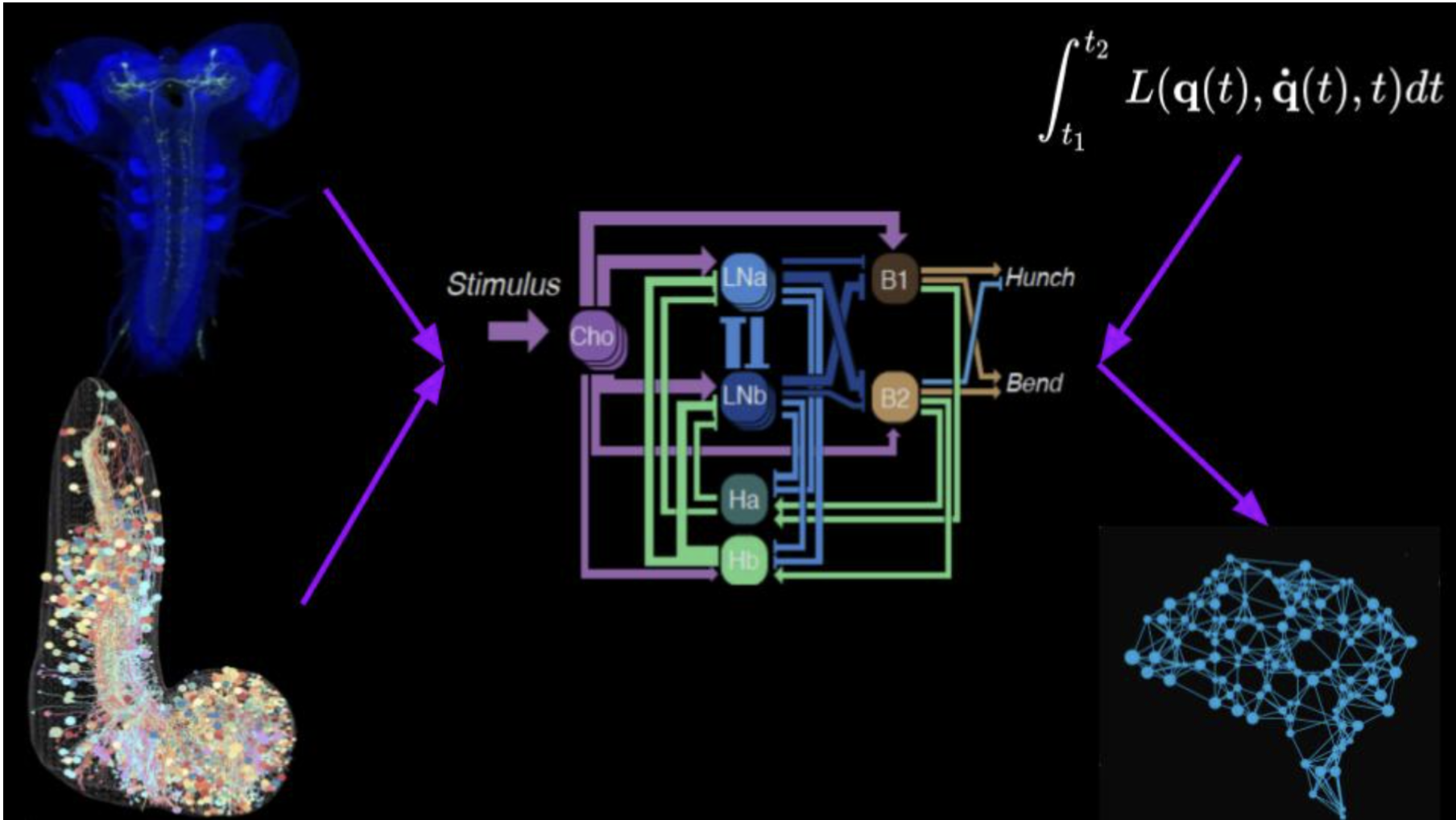
The reason EPIMETHEE has put so much faith in fruit fly larvae is down to the simplicity of their brain structure: where our brains have 100 billion neurons, theirs have only 10,000. But this doesn't prevent them from carrying out complex tasks and adapting to their environment: they are able to crawl, to curl up into a ball if they are attacked, to find food, to react to changes in temperature and to learn certain behaviours.
Jean-Baptiste Masson explains that:
In computer science terms a larva’s brain is a highly compressed system capable of handling uncertainty”. “It is also much simpler and far more versatile than our artificial intelligence tools, with their billions of virtual neurons.
Another benefit of working with larvae is that they cost practically nothing. In the space of a day a drug candidate can be assessed on 100,000 larvae, or even more, while the same operation would take several weeks using 100 or so mice.
Tests are far more instructive when carried out on a large number of subjects, showing a range of responses to the drug. It has been shown that current tests are not reliable enough: in recent years many pharmaceutical companies have had to give up on new drugs after disappointing early results for humans.
An important breakthrough on Alzheimer’s
These findings gave the researchers inspiration for two bold ideas. The first involved exploring, mapping and modelling the biological reality of brain connections within fruit fly larvae brains, before then transposing this knowledge onto human health. The second involved using what is known as ‘bio-inspired’ artificial intelligence in order to create more compact and more adaptable neural networks.
EPIMETHEE was able to make important progress on the first of these thanks to one of its PhD students, Anqi Zhou, who has as one of her supervisors Tihana Jovanic (CNRS). She exposed larvae’s neural circuits to amyloid beta, a peptide thought to be implicated in Alzheimer’s disease, before closely monitoring any changes to these circuits and the consequences these had on larvae behaviour. Jean-Baptiste Masson add that:
We are still some way off developing a model that can be used on humans. But we can show that it is possible to alter neural circuits and demonstrate the effects, accessing the disease directly via these circuits.
Indeed, this search for meaning is central to medical research in neuroscience. Although there are all sorts of imaging methods for analysing the human brain from every possible angle, science is only just beginning to understand this extraordinary machine.
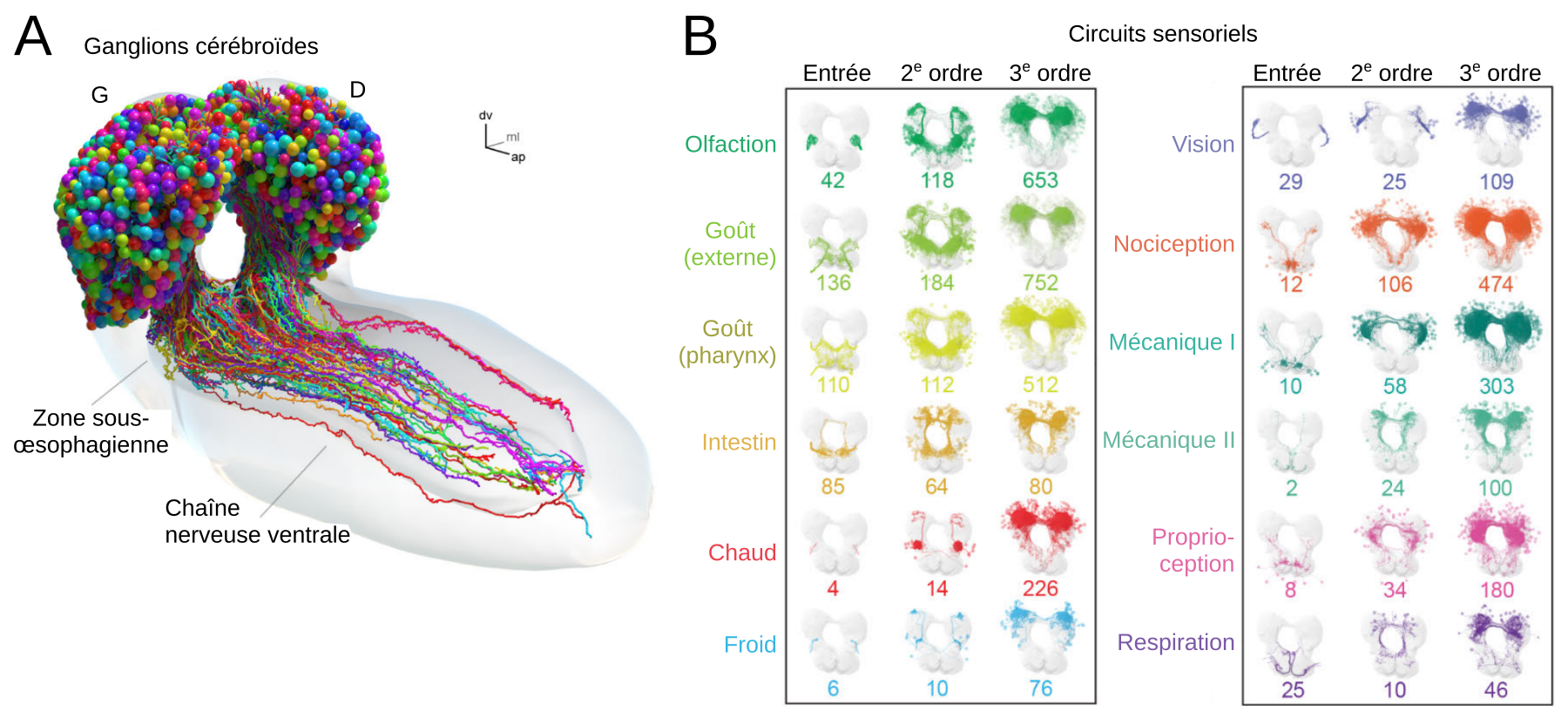
Even within a fruit fly larva’s brain there are multiple levels of complexity. In addition to their primary function, the 10,000 neurons can have either an inhibitory, excitatory or neuromodulatory effect. What’s more, certain biological behaviours can change dramatically as a result of just a handful of neurons.
Mapping the neural connections in order to better link body and brain
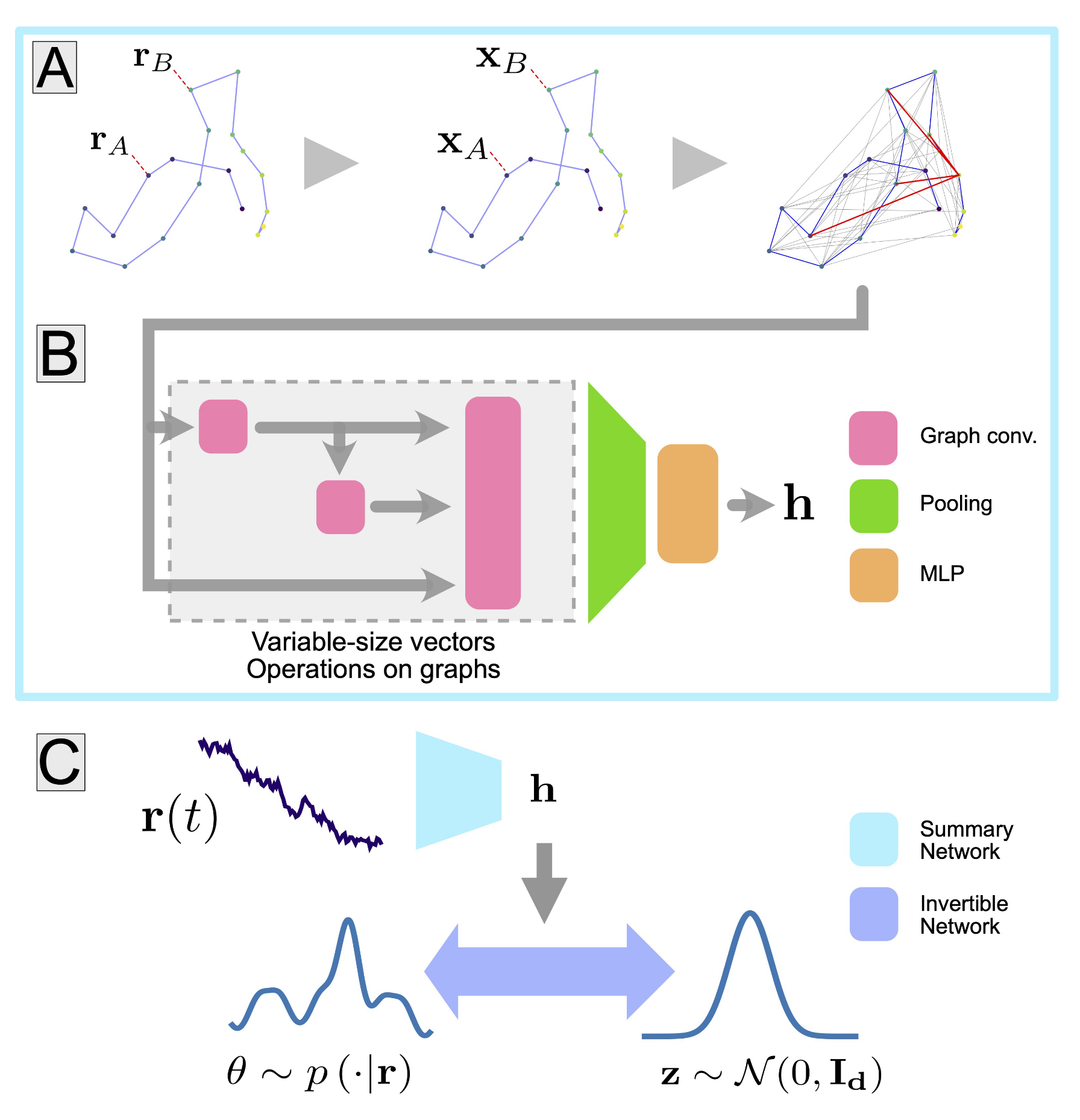
EPIMETHEE’s road map has three stages. The first, which is almost at an end thanks to contributions from a number of different teams, has involved building a map of all of the connections within a larva’s brain: how are the 10,000 neurons all linked to each other? Could this be used to identify and analyse the main neural circuits? The only drawback is that this map is static, and does not show how these circuits mobilise in order to carry out specific tasks, such as locomotion, learning or decision-making.
This correlation will be established during the second stage, which will involve the development of a “virtual larva”, containing both the larva’s brain and body. Jean-Baptiste Masson explains that:
Modelling living things is much harder than creating artificial intelligence tools. To give you an example, larvae can mobilise eleven segments of their bodies depending on what they are doing, one after another or all at the same time, using the same neurons.
This model of the brain's relationship with the body will be crucial in developing applications in human health and carrying out a first full-scale trial within the next five years. The disease it will be used on will depend on the partnerships EPIMETHEEE forms in either academia or industry.
Towards neuroscience-inspired artificial intelligence
Further down the line, the third stage will involve utilising the results from the research on fruit fly larvae to develop a different approach to designing artificial intelligence tools.
One example that Jean-Baptiste Masson has thought of is sensors (for pressure, temperature, vibration, etc.), whose signals - instead of being routed to a remote server - will be processed in record time using a miniaturised artificial intelligence system mounted on the sensor. The aim of this would be to speed up decision-making, whether it's to close a valve, cut off electrical power or to slow down a motor.
Verbatim
Evolution has produced perfectly coordinated decision-making circuits; it's up to us to find other uses for them.
Project team leader
Find out more about EPIMETHEE
- Prix des innovateurs 2022: Jean-Baptiste Masson, founder of Avatar Medical, Île-de-France region, 23/9/2022 (video in French).
- The first complete map of the brain of a fruit fly larva, Science & Vie, 25/3/2023 (in French).
- The neural symphony or the mathematics of the brain, The Conversation, 6/9/2022 (in French).
- Fundamentally Applied: from lab to patient with Jean-Baptiste Masson (video), Pasteur Institute, 28/11/2023 (in English).
- EPIMETHEE project team page on the Pasteur Institute website (in English).